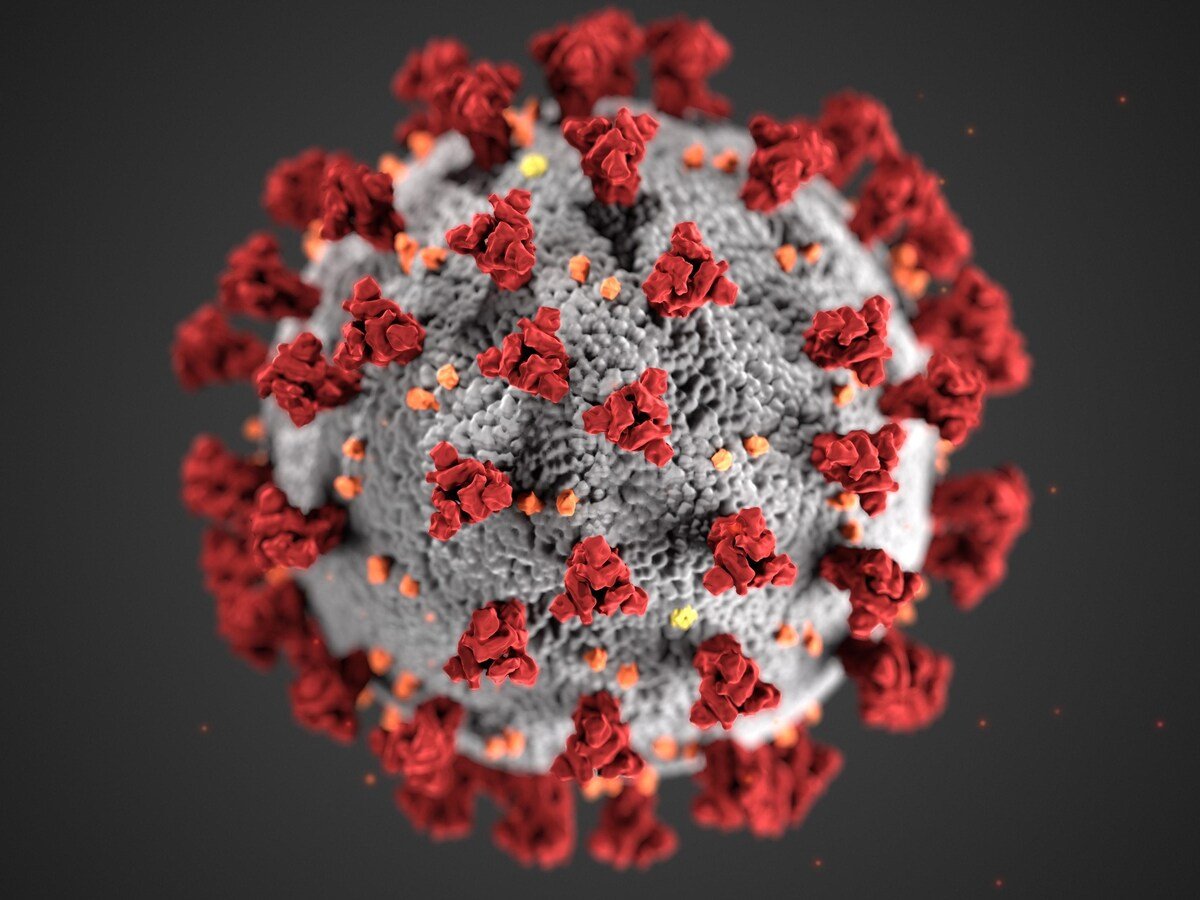
New Delhi, 04 New Delhi, 2025: Scientists have identified a new coronavirus strain, named BRZ batCoV, in moustached bats (Pteronotus parnellii) in Brazil. This discovery marks an important addition to the global coronavirus family tree and broadens understanding of potential zoonotic risks beyond Asia, where most similar viruses have been found so far.
While there is no evidence of human infection yet, experts say the finding underscores the importance of active viral surveillance and continued research into wildlife reservoirs of emerging infectious diseases.
A New Coronavirus Emerges in Brazil
During a recent study of bat populations across different regions of Brazil, researchers collected samples from multiple species to assess the presence of coronaviruses. Genomic sequencing of one particular sample from the moustached bat revealed a previously unknown virus belonging to the betacoronavirus genus — the same family as SARS-CoV-2, which caused the COVID-19 pandemic, and MERS-CoV, responsible for Middle East Respiratory Syndrome.
The newly discovered virus, BRZ batCoV, carries a number of genetic features that drew the attention of virologists worldwide. Among these is a furin cleavage site found at the S1/S2 junction of the virus’s spike protein — a region that plays a key role in helping coronaviruses enter host cells. This feature, which also exists in other highly transmissible coronaviruses, can increase a virus’s ability to infect new species.
Although the sequence and structure of this cleavage site are not identical to that seen in SARS-CoV-2, its presence makes the virus biologically significant and worthy of close monitoring.
Why This Discovery Matters
Bats have long been recognized as natural reservoirs for many viruses, including coronaviruses, Nipah virus, and Ebola virus. They possess unique immune systems that allow them to carry pathogens without developing disease, creating an evolutionary environment for viral mutation and diversification.
The detection of a new coronavirus in South America is particularly significant because most coronavirus surveillance and research have historically focused on Asia and Africa. The discovery of BRZ batCoV suggests that potentially zoonotic viruses exist across continents, and that unmonitored regions may harbor unknown viral threats.
This finding adds weight to the growing understanding that future pandemics could emerge anywhere, not just in historically identified “hotspots.”
Genetic Features of BRZ batCoV
Early genomic analysis has revealed several intriguing features:
- Betacoronavirus Classification – BRZ batCoV belongs to the same subgenus as SARS-related viruses but is distinct from known lineages.
- Furin Cleavage Site – This structural motif in the spike protein may enhance the virus’s ability to infect mammalian cells.
- Unique Genetic Sequence – While related to SARS-like coronaviruses, it shows enough genetic divergence to be classified as a new species.
- Adaptive Evolution Markers – Some parts of the genome indicate potential adaptation to different mammalian hosts, though no direct spillover risk has been confirmed.
These characteristics make the virus scientifically valuable for studying how coronaviruses evolve and adapt in different ecological environments.
Potential Risks and Unknowns
At present, there is no indication that BRZ batCoV can infect humans or domestic animals. However, several factors call for caution:
- Cross-species potential: Genetic features like the furin cleavage site could, under certain conditions, enable the virus to cross species barriers.
- Geographic expansion: The discovery suggests that South American bat species could host a broader diversity of coronaviruses than previously recognized.
- Ecological overlap: Human encroachment into wildlife habitats increases opportunities for virus spillover events.
The scientific community stresses that the finding should not cause panic but should instead prompt stronger global surveillance to monitor emerging viruses before they become public-health threats.
Lessons From Past Outbreaks
The COVID-19 pandemic demonstrated how a virus originating from animals can rapidly spread around the world once it adapts to human hosts. SARS-CoV and MERS-CoV, both coronaviruses with bat origins, have previously crossed into humans via intermediate animal hosts such as civets and camels.
The discovery of BRZ batCoV reminds scientists that viral evolution is continuous and that zoonotic spillovers — though rare — are unpredictable. The most effective prevention strategy is early detection and research transparency, not reactive measures after an outbreak begins.
Expert Insights on Global Preparedness
Virologists emphasize that this discovery should be treated as a scientific alert rather than an emergency. It highlights the need for collaborative, cross-continental monitoring programs that combine field ecology, genomics, and public-health data.
Key recommendations include:
- Expanding wildlife viral surveillance to cover under-studied regions, including South America and Oceania.
- Integrating “One Health” approaches, linking human, animal, and environmental health monitoring systems.
- Investing in rapid genetic sequencing and data-sharing platforms to track virus evolution in real time.
- Reducing human-wildlife conflict, especially in areas where bats and humans share close ecological spaces.
The scientific message is clear: prevention depends on knowledge. The earlier we understand where new viruses originate, the better we can design defenses against them.
The Role of Bats in the Ecosystem
It’s important to remember that bats are vital to the environment. They pollinate plants, disperse seeds, and control insect populations — making them crucial for ecological balance. Demonizing or harming bats would not reduce disease risk; in fact, it could worsen it by disturbing natural habitats and forcing animals into closer contact with humans.
Public health experts advocate for conservation-based prevention, focusing on habitat protection, ecological balance, and minimizing unnecessary human contact with wildlife.
How the Discovery Impacts Pandemic Preparedness
The identification of BRZ batCoV strengthens the argument for continuous and transparent virus monitoring worldwide. Surveillance systems should include routine testing of wild animals, livestock, and environmental samples for potential pathogens.
Laboratories across continents are now being encouraged to share genomic data on new viral discoveries through global databases. This allows scientists to quickly assess similarities, mutations, and potential risks before viruses spread beyond their natural hosts.
The findings also support the idea that pandemic preparedness should not only focus on vaccines and treatment capacity but also on pre-emptive ecology-based research — identifying potential threats before they reach humans.
Should People Be Worried?
At present, there is no reason for public alarm. The virus was detected in wildlife, not in humans, and there is no evidence of transmission outside the bat population.
However, it serves as an important reminder that pandemics do not begin suddenly — they evolve silently in nature long before they reach human society. Keeping global health systems vigilant, funding research, and promoting responsible wildlife interactions remain the best defenses against future outbreaks.
The Broader Message
The discovery of BRZ batCoV comes at a time when the world is still recovering from the COVID-19 pandemic and striving to strengthen health security systems. It highlights that the next pandemic threat could emerge from anywhere, and that proactive surveillance is far more effective than emergency reaction.
This finding encourages a balanced perspective — awareness without fear, and preparedness without panic. Continued research will help determine whether BRZ batCoV poses any risk to humans or if it remains confined to its bat hosts.
As scientists continue to analyze the virus’s genome, they hope to use the data to better understand coronavirus evolution and improve global readiness for any future outbreaks.
The identification of the new BRZ batCoV coronavirus in Brazilian moustached bats is not a cause for alarm but a call for vigilance. It reinforces the understanding that emerging infectious diseases are a global challenge requiring global cooperation.
Every discovery like this adds to humanity’s scientific toolkit, helping predict and prevent future pandemics. Strengthening wildlife monitoring, investing in “One Health” systems, and maintaining transparency across nations will be essential steps in ensuring that the next virus is detected long before it reaches people.
Nature constantly evolves — and so must our vigilance.









Leave a comment