On 26 January 2026, the World Health Organization (WHO) was notified by India’s National IHR Focal Point of two confirmed cases of Nipah virus (NiV) infection in West Bengal, marking the third outbreak of the virus in the state. Both cases involved healthcare workers at a private hospital in Barasat, North 24 Parganas district, and were confirmed by the National Institute of Virology (NIV), Pune on 13 January. One patient remained on mechanical ventilation as of 21 January, while the other, who had experienced severe neurological illness, showed significant improvement. Over 190 contacts were tested with support from a mobile BSL‑3 laboratory deployed by NIV Pune, and all tested negative. No further cases have been reported to date. WHO continues to monitor the situation closely and provides guidance to Indian authorities to contain the outbreak.
What is Nipah Virus?
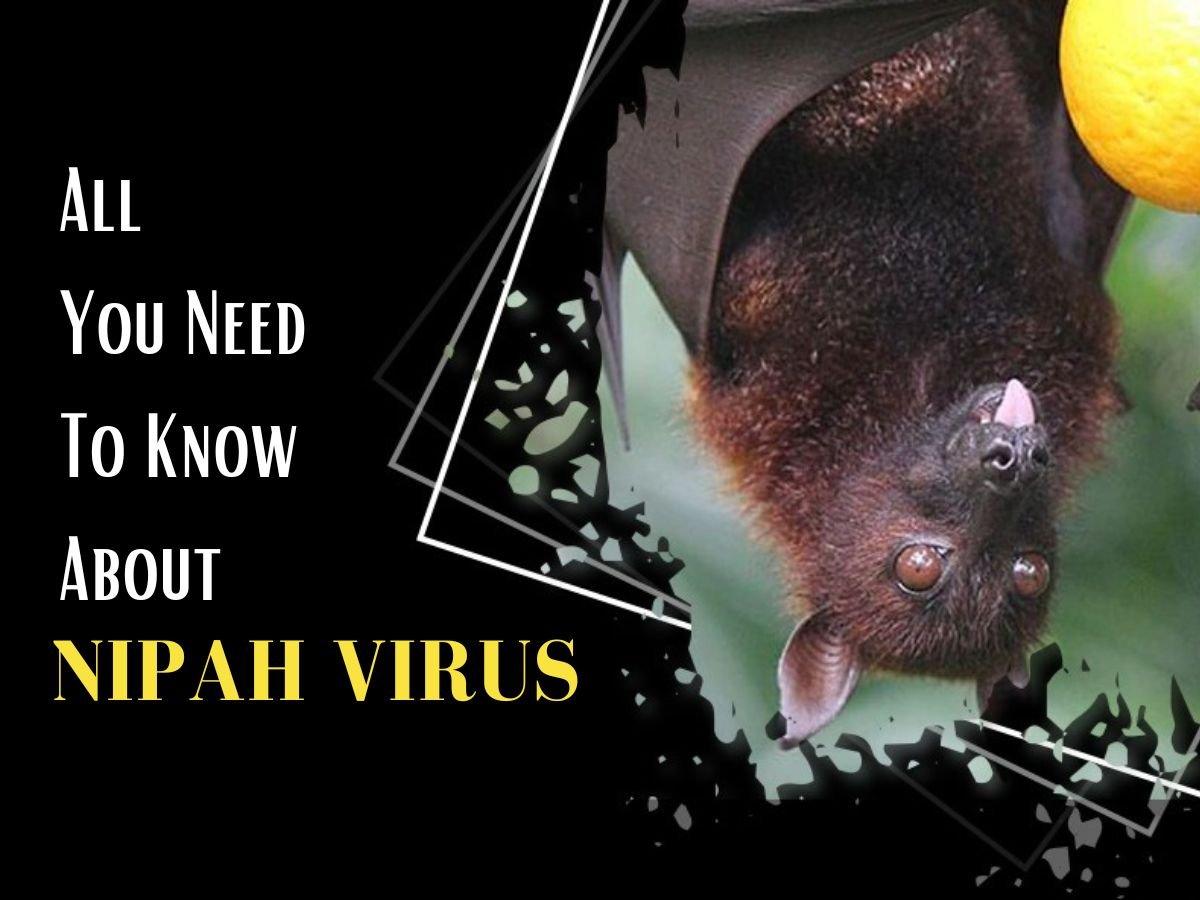
Nipah virus (NiV) is a rare, deadly zoonotic pathogen, primarily carried by fruit bats of the Pteropus genus. According to WHO, NiV can infect humans through:
- Direct contact with infected animals
- Consumption of contaminated food (e.g., raw date palm sap)
- Close contact with infected individuals, including healthcare workers and caregivers
WHO classifies NiV as a priority pathogen due to its high case fatality rate (CFR 40–75%), epidemic potential, and lack of licensed vaccines or specific treatments. Previous NiV outbreaks have occurred in India, Bangladesh, Malaysia, Singapore, and the Philippines, often with limited but severe human-to-human transmission.
The 2026 West Bengal Outbreak
The confirmed cases involved a female and male nurse, both aged 20–30, who developed symptoms typical of severe NiV infection in late December 2025 and were admitted to hospital in early January 2026. Laboratory confirmation by RT-PCR and ELISA was conducted by NIV Pune.
WHO emphasizes that prompt case detection and laboratory confirmation are critical in controlling NiV outbreaks. Contact tracing efforts identified over 190 individuals, all of whom tested negative, demonstrating effective infection prevention measures.
This outbreak is the third in West Bengal, following Siliguri (2001) and Nadia (2007), and the 13th in India overall. WHO continues to support Indian authorities through risk assessment, guidance on infection control, and communication to global public health networks.
How Nipah Virus Spreads
According to WHO, NiV can be transmitted through multiple pathways:
- Animal-to-human transmission: Bats shed the virus in saliva, urine, and feces, contaminating fruits and sap.
- Human-to-human transmission: Close contact with infected patients can transmit the virus via body fluids.
- Foodborne transmission: Consumption of raw or unprocessed products contaminated by bats, such as date palm sap, is a major route in South Asia.
The virus has an incubation period of 3–14 days, though rare cases may extend up to 45 days. Early symptoms are often nonspecific, which can delay diagnosis. WHO underscores the importance of enhanced surveillance and rapid laboratory testing in outbreak settings.
Symptoms and Clinical Presentation
NiV infection ranges from asymptomatic to fatal encephalitis. Initial symptoms, as noted by WHO, include:
- Fever, headache, and muscle pain (myalgia)
- Vomiting and sore throat
- Fatigue and drowsiness
As the disease progresses, neurological and respiratory symptoms can appear:
- Altered consciousness and dizziness
- Seizures and coma within 24–48 hours
- Respiratory distress or atypical pneumonia in some patients
WHO guidance recommends intensive supportive care for severe cases, including mechanical ventilation when necessary, as no antiviral therapy or licensed vaccine exists.
Laboratory Diagnosis
WHO-recommended methods for diagnosing NiV include:
- RT-PCR to detect viral RNA in bodily fluids
- ELISA to detect NiV-specific antibodies
- Virus isolation in BSL‑3 laboratories
India’s deployment of mobile BSL‑3 labs ensures rapid testing, reducing delays in outbreak response.
Public Health Measures in India
India has implemented WHO-aligned public health protocols, including:
- Prompt contact tracing and testing of exposed individuals
- Strengthened surveillance in hospitals and communities
- Health education campaigns to raise public awareness
- Infection prevention and control (IPC) in healthcare settings with WHO-recommended precautions
Healthcare workers handling suspected or confirmed NiV cases follow WHO guidelines, including:
- Use of medical masks, eye protection, gloves, and gowns
- Airborne precautions during aerosol-generating procedures
- Isolation in single-patient rooms
WHO also provides technical guidance to Indian authorities on outbreak monitoring, epidemiological assessment, and risk communication.
Prevention Strategies for the Public
In the absence of vaccines, WHO emphasizes prevention:
- Avoid bat-contaminated food: Boil date palm sap, wash and peel fruits, discard those with bite marks
- Maintain hand hygiene after visiting sick people
- Avoid close contact with infected patients unless using protective measures
- Seek early medical attention for symptoms resembling NiV
- Follow updates from WHO and local health authorities
These measures are particularly important during mass gatherings, where the risk of exposure may increase.
WHO Risk Assessment
According to WHO:
- Sub-national (West Bengal): Moderate risk due to lack of specific treatment and challenges in early diagnosis
- National, regional, global: Low risk as the outbreak is geographically limited and containment measures are in place
- Neighboring countries: Low risk; no cross-border transmission reported
WHO continues to monitor seasonal outbreaks linked to bat activity and cultural practices such as date palm sap consumption.
Historical Context of NiV in India
- 2001 Siliguri, West Bengal: 66 cases
- 2007 Nadia, West Bengal: Limited cases, well contained
- 2018 Kerala: 18 cases
- Outbreaks usually occur December–May, aligning with bat activity and sap harvesting
WHO notes that while the number of NiV cases remains low, repeated spillover events highlight the need for ongoing vigilance and preparedness.
Research and Future Preparedness
WHO has classified NiV as a priority pathogen under its R&D Blueprint, emphasizing the need for:
- Vaccine development
- Antiviral therapies
- Rapid diagnostic tools
- Strengthened One Health approaches integrating human, animal, and environmental surveillance
India has developed rapid response teams, advanced laboratories, and surveillance networks to respond to outbreaks, demonstrating the ability to contain NiV effectively.
The 2026 Nipah virus outbreak in West Bengal underscores the importance of early detection, supportive care, and preventive measures. While rare, NiV infections are highly fatal, and WHO guidance remains central to outbreak management. Public awareness, healthcare preparedness, and adherence to preventive measures can reduce the risk of transmission and safeguard communities.
India’s experience, combined with WHO technical support and global monitoring, ensures that the 2026 outbreak is effectively contained, while lessons learned continue to strengthen future pandemic preparedness.