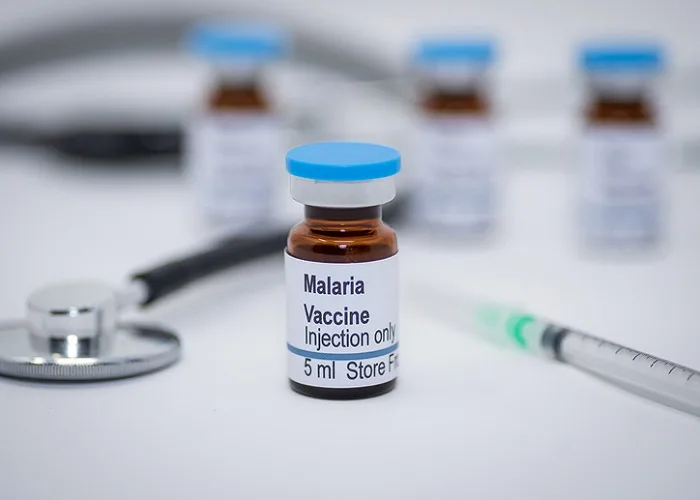
New Delhi, 21 July 2025: India’s premier research body has launched a major initiative to develop AdFalciVax, an indigenous multi‑stage malaria vaccine, as part of its malaria elimination strategy. The vaccine is being created by a consortium led by a regional research centre, with support from national immunology and malaria research institutes. Designed as a recombinant chimeric vaccine, it targets two critical stages of the Plasmodium falciparum parasite—aiming to both prevent human infection and reduce community transmission.
What Makes AdFalciVax Unique
Unlike existing malaria vaccines that target a single parasite stage, this vaccine is engineered to attack both the pre‑erythrocytic (infection) and sexual (transmission) phases. Its antigen components are produced using Lactococcus lactis, a safe food‑grade bacterium, enabling dual protective mechanisms. Preclinical trials have shown excellent efficacy, reduced risk of immune evasion, potential for stronger long‑term immunity, and thermal stability at room temperature for more than nine months—a major logistic advantage in tropical and remote areas.
Preclinical Results and Potential Benefits
In early lab testing, the vaccine demonstrated broader protection compared to existing formulations like RTS,S/AS01 (Mosquirix) and R21/Matrix‑M. Strengthened antibody responses, lower risk of parasite escape, and sustained activity post-boost in animals suggest the possibility of enhanced long-term immunity. Its extended shelf-life under standard conditions reduces dependency on cold-chain infrastructure, making it ideal for widespread use in low-resource communities.
Development Timeline & Licensing Plan
The technology is still in the preclinical stage; human trials—including Phase I—are expected to begin within the next two years. The complete development, including clinical testing and regulatory clearance, is projected to take approximately seven years across four structured phases, with six-month buffers built into each stage.
The research body has issued an Expression of Interest (EoI) inviting Indian companies and vaccine manufacturers to participate in non-exclusive technology transfer agreements. Selected organizations will be licensed to refine, manufacture, market, and distribute the vaccine, while research institutes will continue to provide technical support and expertise throughout the development and production process. Royalty terms and intellectual property provisions are defined under the institutional policy.
Strategic and Public Health Implications
If successful, AdFalciVax could become one of the world’s most advanced malaria vaccine candidates, aligning with national priorities like the “Make in India” initiative and malaria elimination targets by 2030. It aims to complement existing vaccine options—such as Mosquirix and R21/Matrix–M—for children in high-burden settings. Its dual-stage targeting and easier storage could also make it more effective and accessible across regions that struggle with cold-chain logistics.
Summary Table: AdFalciVax at a Glance
| Feature | Current Info |
|---|---|
| Type | Indigenous recombinant chimeric malaria vaccine (dual‑stage) |
| Target Parasite | Plasmodium falciparum |
| Development Partners | Regional Medical Research Centre, Malaria Research Institute, National Institute of Immunology |
| Key Advantages | Broader protection, risk reduction of immune evasion, long-term immunity, heat stability |
| Storage | Stable at room temperature ≥ 9 months |
| Development Timeline | ~7 years (preclinical to commercialization) |
| Licensing Model | Non‑exclusive tech transfer to multiple manufacturers |
| Support Mechanism | Technical guidance from research institutes |
| Public Health Role | Complements existing WHO-backed vaccines; aids elimination efforts |
According to the researchers, AdFalciVax has the potential to revolutionize India’s approach to malaria prevention, with broad implications for global public health. It reflects the country’s growing leadership in vaccine innovation and its commitment to equitable access in malaria-endemic settings. ICMR’s focus now shifts to securing collaborations, initiating human trials, and scaling production under robust regulatory and IP frameworks.









Leave a comment